Dr.-Ing. Revathi Appali
Junior Research Group Leader and Principal Investigator
Academic Mentor
SFB 1270/2 ELAINE – Electrically Active Implants
Raum Ex 139
Tel.: +49 (0) 381 498-7071
Fax: +49 (0) 381 498-7081
E-Mail: revathi.appali(at)uni-rostock.de
Homepage: http://www.revathiappali.sitew.de

Research Group Members
| Doctoral Researchers | Post-doctoral Researchers | Student Assistants | Interns |
|---|---|---|---|
| M.Sc. Vishnu Prathapan | Dr. Karthik Sridhar | N.N. | M.Sc. Shubhangi Goyal |
| M.Sc. Jan Philipp Payonk | Dr. Pavel Arnold Ndjawa Yomi |
Co-Supervision
| Doctoral Researchers |
|---|
| M.Sc. Lam Vien Che |
Past members
| Post-doctoral Researchers | Student Assistants |
|---|---|
| Dr. Anna Karina Fontes Gomes | Saravana Subramani |
| Dr. rer. nat. Jonathan Dawson | |
| Dr.-Ing. Kiran Kumar Sriperumbudur |
July 2021: Ranked in the list of W1 Professor for Theoretical Electrical Engineering of Technical Faculty of Christian-Albrechts-University of Kiel
07/2021 to 06/2025, Co-Principal Investigator of Project A02 in SFB 1270/2 ELAINE
Keywords: Multiscale mathematical modeling, Coupled problems, Deep Brain Stimulation, Osseoinduction, Uncertainty Quantification
02/2019 to 06/2025, Junior Research Group Leader in Project A02 of SFB 1270/1 ELAINE
Keywords: Multiscale mathematical modeling, Coupled problems, Deep Brain Stimulation, Osseoinduction, Uncertainty Quantification
11/2014 to 01/2019, Principal Investigator, Deutsche Forschungsgemeinschaft (DFG) - Project number 260482124
Investigation of Electro-mechanical Coupling in Neuronal Membrane - Towards an Electro-mechanical Model of Nerve Pulse Propagation
Keywords: Multiscale mathematical modeling, Coupled problems
04/2013 to 10/2014, Research Associate
Uncertainty quantification in neuron models, DFG proposal preparation,
Co-supervising an external PhD student
03/2009 to 03/2013, Doctoral Researcher in Research Training Group GRK 1505 welisa
Dissertation on Modeling the coupling of action potential and electrodes
Supervisors: Prof. Dr. Ursula van Rienen, Prof. Dr. Jan Gimsa
03/2008 to 11/2008, Master Thesis Student
Development and simulation of four wheel parallel hybrid drive
IAV GmbH, Gifhorn, Germany
Supervisors: Dr. Thomas Boehme, Prof. Dr. Bernhard Lampe
04/2007 to 03/2008, Student Assistant
Data analysis and automation of western blot experiments
Systems Biology Group, University of Rostock
Supervisors: Dr. Katja Rateitschak, Prof. Dr. Olaf Wolkenhauer
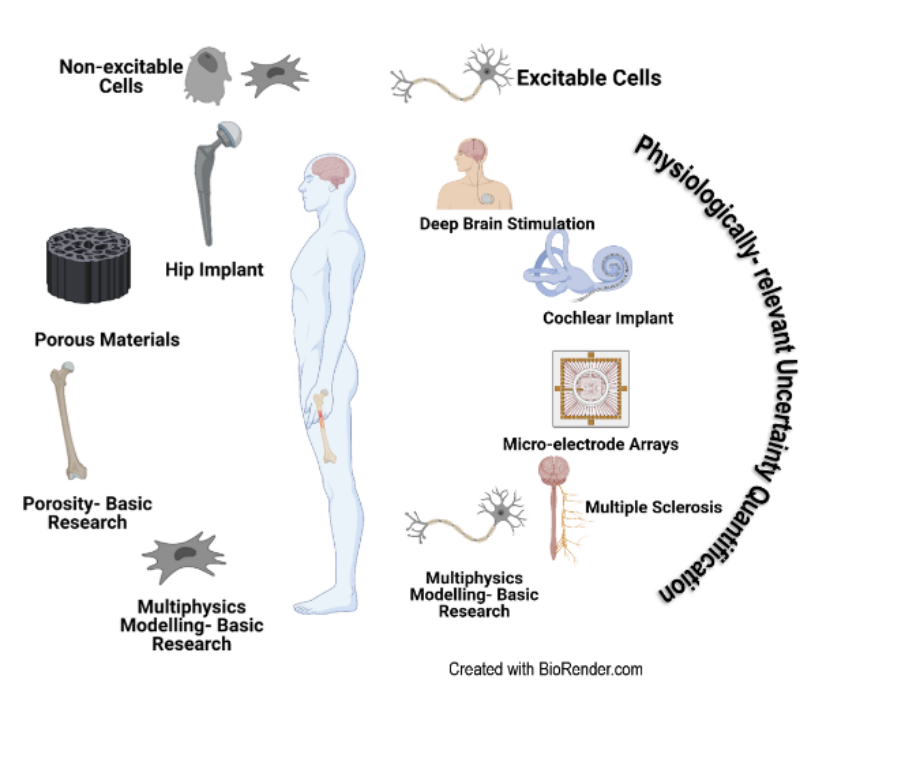
- Modeling and simulation of biological systems for implant studies and their interactions
- Deep Brain Stimulation
- Bone implants
- Cochlear implants
- Biological neural network modeling
- Physiologically relevant uncertainty quantification in electrically active implants
- Multiphysics modeling of
- Stem cell differentiation and proliferation in electrical fields
- Neuronal signal generation and propagation
- Multiscale modeling of
- Bone cell differentiation and proliferation in a bioreactor
- Deep brain stimulation for Parkinson’s disease and Dystonia
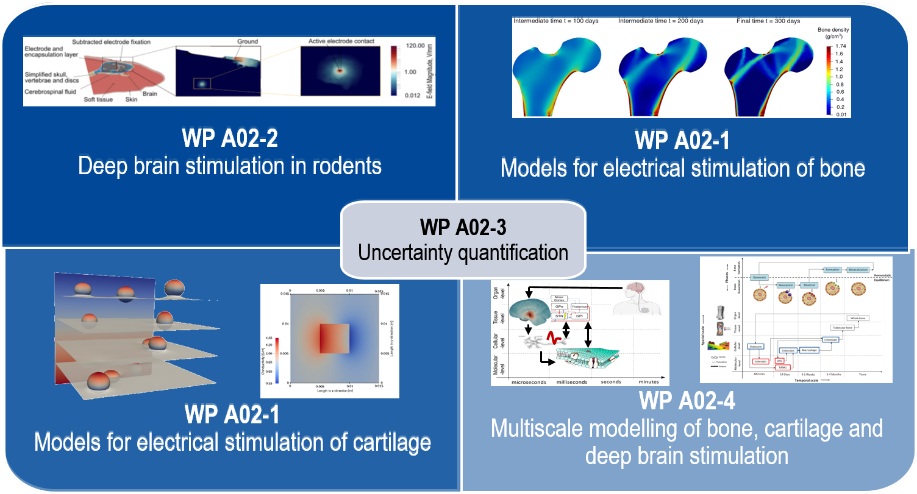
Project A02 in SFB 1270/2 ELAINE – ELectrically Active ImplaNts
Project A02 : Multi-scale models for studies on electrically active implants in due consideration of uncertainties in the input data

Project A02 investigates microscopic and macroscopic simulations in continuous media in context with the therapeutical electrical stimulation of bone, cartilage or deep brain structures.
Essential is the numerical solution of the relevant partial differential equations (Maxwell’s equations, etc.) with the finite element method to determine the desired quantities, particularly the electric field distribution. For the electrical parameters of the biological tissues, primarily parameterized relaxation models are employed. As already started on a pilot basis in the first funding period of the Collaborative Research Centre, values from project A05 can be used in the project. Electrical and electrochemical effects at the electrode-tissue interface, which depend on the selected type of stimulation, are employed through equivalent circuit models. In the first funding period, suitable theoretical and numerical models were selected. They will be further extended or refined. Results from simulations on a smaller scale are incorporated via homogenization approaches, equivalent circuit models or parameter extraction. Besides extending the multiscale and multiphysical models, establishing stochastic and inverse methods for determining patient-specific optimized recommendations for stimulation parameters presents another focus for the second funding period. To this end, an open-source, multipurpose simulation platform based on the tissue-specific solutions established in the first funding period will be created. The clinical researchers in the consortium will test the simulation platform utilizing easy-to-use graphical user interfaces.
Project duration: 01.07.2021 to 30.06.2025
Principal Investigators: Prof. Dr. Ursula van Rienen, Dr.-Ing. Revathi Appali
Team involved: M.Sc. Jan Philipp Payonk, M.Sc. Lam Vien Che, Dr. Anna Karina Fontes Gomes
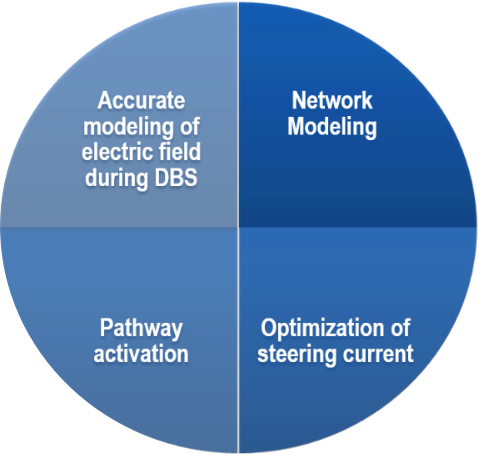
Project S01 in TRR 295 ReTune: Treatment of motor network disorders using neuromodulation

Deep Brain Stimulation (DBS) is a well-accepted treatment option for movement disorders such as Parkinson’s disease. Since DBS target structures are small, exact positioning of surgically-implanted electrodes is crucial to yield optimal outcomes. However, the most effective target position for DBS electrodes is still under debate. To study the relationship between clinical outcomes and electrode placement across patients, the field of DBS imaging evolved. This service project will be based on a widely-used software tool called Lead-DBS, which enables precise localization of DBS electrodes and modelling of their therapeutic effects. Our two primary goals are first, to perform electrode localizations for other projects within the TRR and second to further enhance Leas-DBS and our methodology in DBS modelling. This will involve two research projects, namely (i) a pipeline for multimodal imaging and precise subcortical segmentation of target structures, and (ii) advanced modelling of electric fields and the neural activation around the electrode to study the impact of DBS on surrounding tissue. These efforts will significantly improve imaging methodology for DBS and offer a support structure to answer the central research questions of the TRR 295.
Project duration: 01.07.2020 to 30.06.2024
Principal Investigators: Prof. Dr. Ursula van Rienen, Dr. med. Anna Tietze, Prof. Dr. med. Andreas Horn
Team involved: M.Sc. Arash Golmohammadi, Dr.-Ing. Revathi Appali
Investigating the synergistic effects of spatially resolved biochemical, physicochemical, and physical key stimuli to generate biomimetic niches in perfusion bioreactor and their proficiency to derive large bone-like constructs.
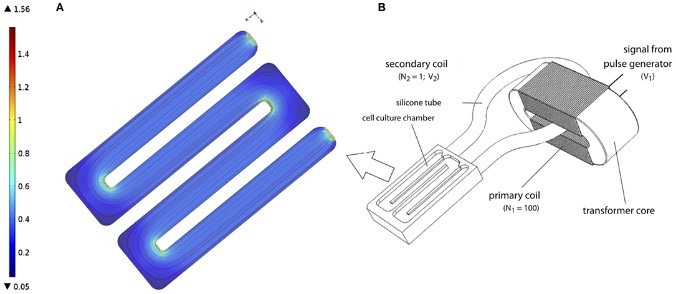
Bioreactors are used increasingly in bone tissue engineering approaches, but the existing homogenous niche applied is generally insufficient to achieve/support formation of bone-like constructs. In physiology, multiple osteochondral cells are present in high cell density and their dynamic interactions are critical for bone development and regeneration processes. This is also complemented with supporting parameters such as oxygen tension (pO2) distribution, biochemical stimuli, and physical/mechanical stimuli to establish the dynamic niches essential for the growth of naïve organs (i.e. cartilage and bone) and attaining their functionalities. In this proposal, we aim to investigate the synergistic potentials of 5 distinct parameters to recapitulate bone developmental processes in a perfusion bioreactor. Through the established dynamic biomimetic niches, the effects on construct development and capability to achieve bone-like tissues in vitro will be assessed. These parameters include 1) medium pH; 2) pO2; 3) Young´s modulus of 3D-printed scaffolds; 4) multi-step osteochondral differentiation regimes and 5) Transformer-like induced electric field (TLC-EF) stimulation to emulate piezoelectric stimuli during movement. Human bone marrow-derived mesenchymal stem cells (hMSC) and human induced pluripotent stem cells (hiPSC) will be used to define the proficiency of biomimetic niches. Their potential to derive different osteochondral cell-types in the biomimetic niches will be critically assessed. Specifically for TLC-EF stimulations, multiscale modelling/simulations that describe EF/Bone construct interactions and the effects on osteogenesis will be combined with experimental validations. This is done with the aim to define an effective range of EF parameters for bone tissue engineering applications. Ultimately, this study will result in a specific set of conditions/stimuli that can recapitulate physiological-like niches in vitro. The approaches applied will also introduce a novel concept to the field of tissue engineering and can be further exploited to derive bone-like constructs with near-physiological properties in a bioreactor.
Project duration: 3 Years (from 07/2021)
Principal Investigators: Prof. Dr. Ursula van Rienen, Dr.-Ing. Poh Soo Lee, Dr.-Ing. Benjamin Kruppke
Team involved: Post-doctoral researcher (Uni Rostock), Dr.-Ing. Revathi Appali
TU Dresden: Dr.-Ing. Poh Soo Lee (Eigene Stelle), PhD Student: Dipl.-Ing. Franziska Alt – supervised by Dr.-Ing. Benjamin Kruppke
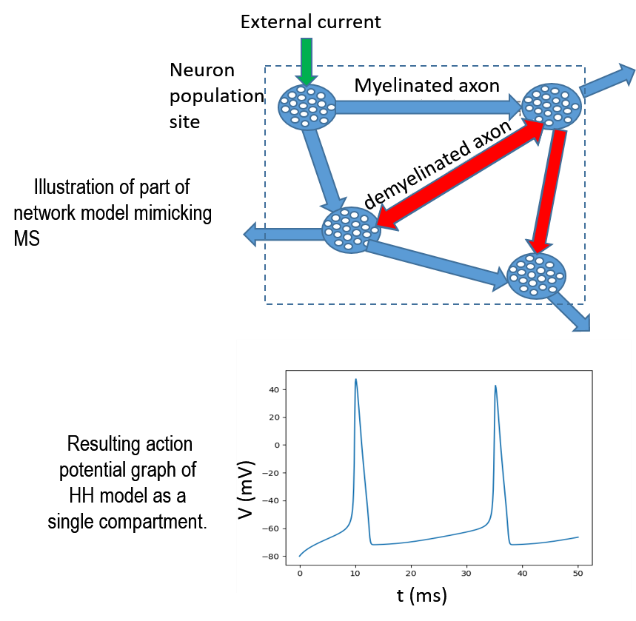
Predicting Spreading of Multiple Sclerosis Demyelination Through a Trans-synaptic Modeling Approach

This project aims at understanding the effects of demyelination and remyelination in Multiple Sclerosis through a trans-synaptic modelling approach. We use neuroVIISAS (https://neuroviisas.med.uni-rostock.de/neuroviisas.shtml) developed by Prof. Dr. Oliver Schmitt to carry out this study. neuroVIISAS (neuro Visualization, Imagemapping, Information System for Analysis and Simulation) is an open framework for integrative data analysis, visualization and population simulations.
Project duration: 2019 – 2021
Supervisors: Prof. Dr. Ursula van Rienen, Prof. Dr. Oliver Schmitt, Dr.-Ing. Revathi Appali
State Graduate Fund Holder: M.Sc. Vishnu Prathapan
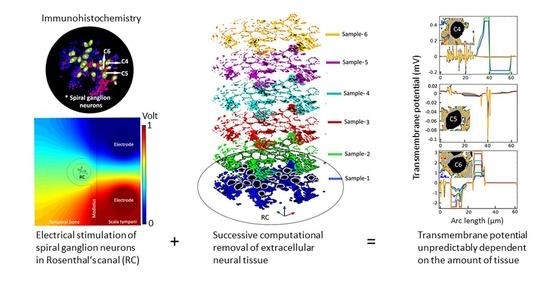
Modeling the effects of morphological and physiological parameters on the electrical stimulation of the auditory nerve by cochlear implants
Sensorineural deafness is caused by the loss of peripheral neural input to the auditory nerve, resulting from peripheral neural degeneration and/or a loss of inner hair cells. Provided spiral ganglion cells and their central processes are patent, cochlear implants can be used to electrically stimulate the auditory nerve to facilitate hearing in the deaf or severely hard-of-hearing. Neural degeneration is a crucial impediment to the operational success of a cochlear implant. The present, first-of-its-kind two-dimensional finite-element model investigates how the depletion of neural tissues might alter the electrically induced transmembrane potential of spiral ganglion neurons. The study suggests that even as little as 10% of neural tissue degeneration could lead to a disproportionate change in the stimulation profile of the auditory nerve. This result implies that apart from encapsulation layer formation around the cochlear implant electrode, tissue degeneration could also be an essential reason for the apparent inconsistencies in the functionality of cochlear implants.
Team Lead: Prof. Dr. Ursula van Rienen
Team involved: Dr.-Ing. Kiran Sriperumbudur, Dr.-Ing. Revathi Appali
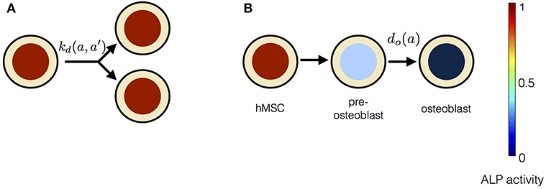
Effect of Electric Field on Stem cells and Bone cells
This project aims at studying the effect of electric field stimulation on human stem cells and bone cells. We use data driven mathematical modelling as a tool to understand the underlying phenomena.
Team lead: Dr.-Ing. Revathi Appali, Prof. Dr. Ursula van Rienen
Team involved: Dr. rer. nat. Jonathan Dawson, Dr.-Ing. Revathi Appali
Uncertainty Quantification in Deep Brain Stimulation and Osseoinduction
We are developing physiologically based uncertainty quantification regarding main model parameters. The aim is to optimize the functionality of deep brain stimulation and osseoinduction.
Team Lead: Dr.-Ing. Revathi Appali, Prof. Dr. Ursula van Rienen
Team involved: Dr.-Ing. Kiran Sriperumbudur, Dr.-Ing. Revathi Appali
Investigation of Electro-mechanical Coupling in Neuronal Membrane
Study of Electrical Action Potential in Neurons
This project aims to address different factors that influence the action potential in neurons and its activation in any electrically active implants. We focus on the morphology of neurons and the density of neurons in the region of interest as significant factors involved in the optimized stimulation of neurons.
Team Lead: Prof. Dr. Ursula van Rienen, Prof. Dr. Christiane Thielemann
Team involved: Dr.-Ing. Robert Bestel, Dr.-Ing. Revathi Appali
- Computational Modelling of Physiological Systems (offered in winter semester)
Teaching Assistant:
- Laborpraktikum Theoretische Elektrotechnik 1 und 2
- Advanced Computational Electromagnetics and Multiphysics
- Advanced Electromagnetic Simulation and Multiphysics
Teaching Substitution:
- Theoretische Elektrotechnik 1 in WS 2020/21 during sabbatical of Prof. Dr. Ursula van Rienen
Unique IDs
- ORCID: orcid.org/0000-0001-9668-8185
- Scopus Author ID: 36536600500
- Google scholar link: Revathi Appali